When determining what the reaction is, keep in mind that there has to be a driving force that causes an equilibrium shift. When two atoms in these columns get together, expect to see a few multiple bonds oxygen and nitrogen love forming double and triple bonds when they can, and carbon will oblige with these two and a few others, but typically prefers single bonds with other carbons and most other nonmetals (allowing it to form those long chains that create the backbone of life). You can compute a balanced bond structure using valence electrons, much like calculating ionic charges oxygen needs two valence electrons and so it will form no more than two bonds with other elements. A double or triple bond is very strong, while certain other combinations are strong for other reasons (bonds between carbon and the "Big 3" highly electronegative elements, namely oxygen, fluorine and chlorine, are commonly very strong). How strong these bonds are depend on what elements are being bonded and how many electrons each is sharing with its neighbor. The lighter the compound, the more it wants to gain an electron, with fluorine being the most aggressive electron receptor in the table (to the point where it will try to take electrons from other elements that don't want to give them up, like other halogens and noble gases).Ĭolumns between the halogens and transition metals (from left to right, the boron group or triels, the carbon group or tetragens, nitrogen group or pnictogens, and oxygen group or chalcogens) are more of the "sharing" type they'll take electrons outright from the alkalis, and if they really have to they'll give them up to a halogen like fluorine or chlorine, but they prefer a "give-and-take" relationship, and form shared-orbital "covalent bonds" with most other elements of these groups and with the transition metals. The elements in the halogen group (fluorine, chlorine, bromine, iodine) like to gain electrons, especially from the alkalis. The alkali metals, in the 1 and 2 groups, usually want to lose their one or two valence electrons the heavier the alkali metal, the more the atom wants to lose the electrons, with cesium being the most reactive alkali metal that has a non-radioactive isotope. Cations are then attracted to anions, and these charges must balance a +2 cation such as Mg 2+ or Ca 2+ requires one -2 anion (carbonate, sulfate, hydrogen-phosphate) or two -1 anions (chloride, fluoride, bicarbonate). When they gain them, they become negative anions, and then they lose them, positive cations. When atoms outright gain or lose electrons, they become ions, and gain a charge based on the difference between the protons and electrons in their configuration. The transition metals are "stable" in various valence configurations based on their more complex d-orbital valence structure (the s and p orbitals exposed by the first two and last six columns of the table are more predictable). For elements not in the "transition metal" groups, this is pretty straightforward elements to the left of the transition metals generally want to lose electrons to empty their valence shell (acquiring the stable configuration of the next lightest noble gas), while elements to the right of the transition metals want to share or gain electrons to fill their valence shell, acquiring the configuration of the next heaviest noble gas. Most elements do not have a stable valence shell, and so will want to react in ways that increase their stability.

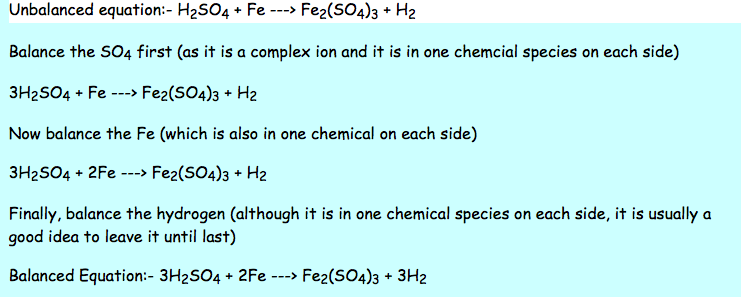
For that, remember the concept of valence shells the number of electrons in the outermost "shell" of its orbitals. What can change in a reaction is the number of electrons each atom "owns". No strictly chemical reaction will cause a carbon to become a nitrogen by gaining a proton. Therefore, every atom of every element on the left side of the arrow has to appear on the right side, with all their protons and neutrons intact.

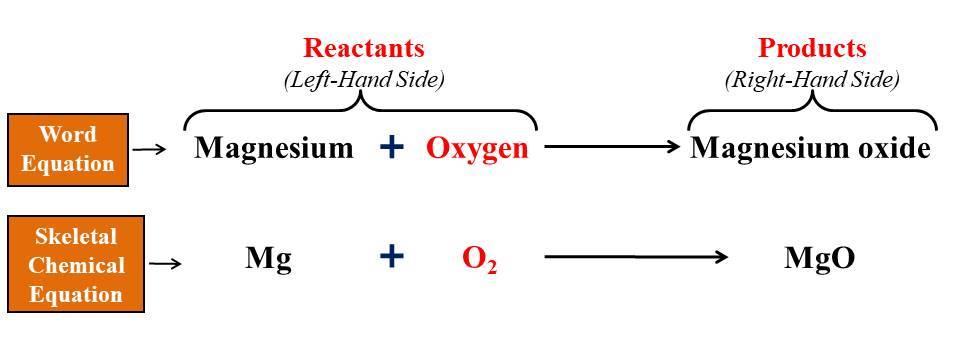
OK, here goes a semester of college chemistry in one post.įirst off, remember the law of conservation of matter in chemical reactions, matter is neither created nor destroyed.
